The curling wheeze and tight-lipped breathing of inflammatory lung conditions (1), the severe abdominal pain and gastrointestinal disruption of inflammatory bowel disease (IBD) (2), and the arduous stiffness associated with rheumatoid arthritis (RA) (3) are common inflammatory ailments that globally prevail. Though the aetiology of each disease is not well understood, each share a similar pathophysiology of unaccounted immune cell recruitment and inflammation (1, 2, 3). With this gap in knowledge, current therapies aim at managing symptoms and preventing further damage by targeting inflammatory pathways (1,2,3). However, the toxicity associated with immune modulators has created an urgency into finding alternative therapies.
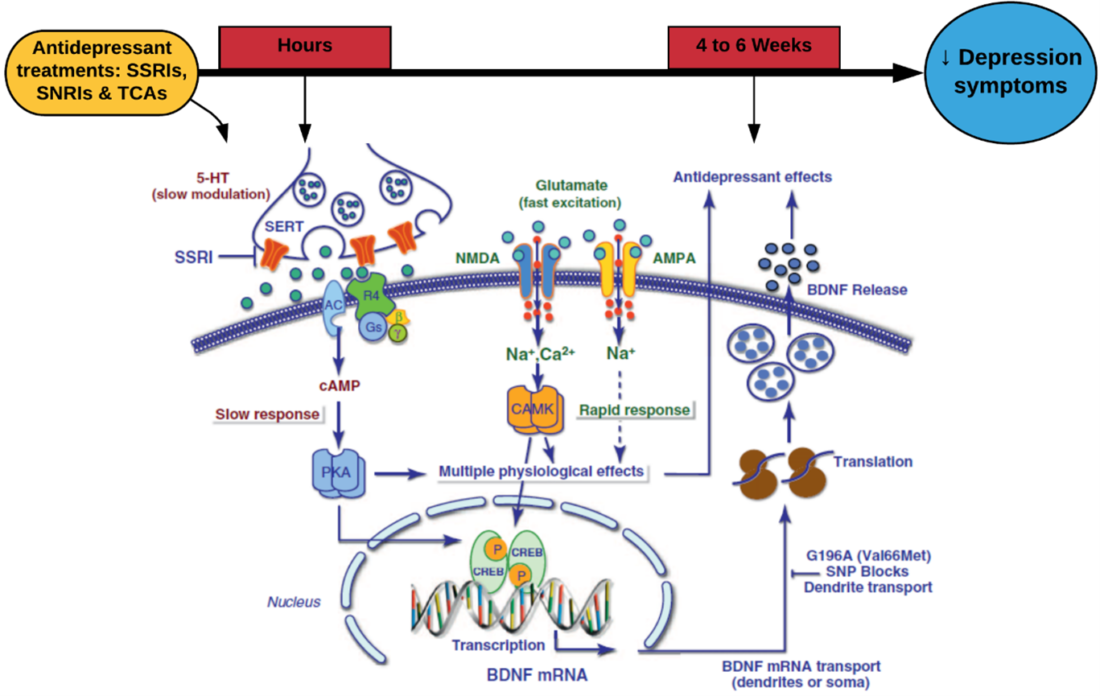
Recently, Incannex Healthcare received approval for a phase I study investigating the safety profile of IHL-675A, a novel anti-inflammatory that is indicated for the treatment of inflammatory diseases including inflammatory lung conditions, IBD, and RA (4). IHL-675A is comprised of cannabidiol (CBD) and hydroxychloroquine (HCQ) (4). Currently, CBD has only been approved for the treatment of seizure disorders (Lennox-Gastaut syndrome and Dravet syndrome) (5).
Over the years, the importance of the endocannabinoid system in controlling homeostasis and mediating metabolic and inflammatory pathways has become a popular target for pharmacotherapy (6). CBD is one of the main active cannabinoids found in Cannabis sativa (5). Cannabinoids are an exogenous molecule that share structural and chemical similarities to cannabinoids (6). Though the mechanism of action remains elusive, pre-clinical studies have shown that CBD indirectly improves anti-inflammatory pathways by inhibiting T cell proliferation, inducing T cell apoptosis, and reducing the migration and adhesion of immune cells (6).
Conversely, HCQ is a well-tolerated anti-malarial and anti-inflammatory compound that has been traditionally indicated for the treatment of RA and malaria (7). Despite the anti-inflammatory mechanism remaining unclear, studies have shown that HCQ disrupts antigen-presentation and processing, preventing activation and differential of CD4+ T cells (7). Furthermore, studies have shown that HCQ induces apoptosis of autoreactive T cells, and interferes with the activation of the humoral immune response, thus reducing the presence and production of inflammatory cytokines (7).
Despite both compounds have individually shown anti-inflammatory properties, preclinical studies conducted by Incannex into IHL-675A has demonstrated that CBD and HCQ act synergistically to inhibit the production of inflammatory cytokines (IL-1, Il-6, TNFα, IL-12, and IFN-γ) in mouse models (7). Furthermore, IHL-675A demonstrated greater anti-inflammatory properties compared to CBD by 109% to 767% from 24 hours post-drug administration (7). These preclinical studies, though minimal, show promising results for the treatment of inflammatory conditions in humans.
Though the hurdle of a phase I trial is hard to surpass, the idea of a safe and effective treatment provides silver-lining to the treacherous battle of chronic inflammatory conditions. The competitiveness of the pharmaceutical industry is the foundation of innovative drug developments. IHL-675A has shown to be a strong competitor within this battle, and hopefully within a few years, a solution to inflammatory conditions is approved.
References:
- Agarwal A., et al., Chronic Obstructive Pulmonary Disease, 2022. StatPearls Publishing, Available at https://www.ncbi.nlm.nih.gov/books/NBK559281/
- McDowell C., et al., Inflammatory Bowel Disease, 2022. StatPearls Publishing, Available at https://www.ncbi.nlm.nih.gov/books/NBK470312/
- Chauhan K., et al,m Rheumatoid Arthritis, 2022. StatPearls Publishing, Available at https://www.ncbi.nlm.nih.gov/books/NBK441999/
- SmallCaps, Incannex Healthcare to begin phase 1 clinical trial for IHL-675A following ethics approval, Nicholaos L., 2022, Available at https://smallcaps.com.au/incannex-healthcare-phase-1-clinical-trial-ihl-675a-following-ethics-approval/ [Accessed 11AUG2022]
- Meissner H., et al., Cannabidiol (CBD), 2022. StatPearls Publishing, Available at https://www.ncbi.nlm.nih.gov/books/NBK556048/
- Atalay S., et al., Antioxidative and Anti-Inflammatory Properties of Cannabidiol. Antioxidants, 2020; 9(1) 20
- Bansal P., et al., Hydroxychloroquine: a comprehensive review and its controversial role in coronavirus disease 2019. Annals of Medicine, 2021; 53(1): p. 117-134
- Incannex, Positive In vivo results confirm strong synergistic activity of IHL-675A to inhibit inflammation, Incannex, 2020, Available at https://company-announcements.afr.com/asx/ihl/cf6be2f2-2d2b-11eb-8f4b-aa3736f8de15.pdf [Accessed 11AUG2022]